Enema Device for Children with Spina Bifida
Overview
The goal of the project is to improve the quality of life for children and adults with fecal incontinence by developing an enema administration device that improves upon current methods. The largest affected group are children with spina bifida. Spina bifida is a birth defect of the neural tube that hampers the development of the brain and spinal cord, often resulting in fecal incontinence. Patients with a neurogenic bowel require a daily enema to evacuate stool. Current administration is carried out with a Foley catheter. The Foley catheter contains a balloon that inflates inside the anal cavity to provide a seal. This balloon is only available in 30 mL and 60 mL sizes, which can leave patients with reduced sphincter tone vulnerable to the catheter falling out during enema administration. The goal is to redesign the administration with an adaptable device that ensures a clean, leakage-free administration. The proposed solution is a supportive device to supplement administration with the Foley. The device forms a flange connection between the Foley and itself, which is maintained by a belt and strap system the user wears. The client for this project is Dr. Andrea Bischoff, a pediatric surgeon at Children’s Hospital Colorado Anschutz Medical Campus.

Team Members
- Ryan Croghan
- Mary King
- Rachel McManus
- James McNamara
- Gabriel Muehlbauer
The Client
- Children’s Hospital Colorado
Acknowledgements
Project Advisor: Donna Bodeau
Technical Advisors: Damian Friend and Judith Klein
Project Sponsors: Children’s Hospital Colorado, Dr.Bischoff, Dr.Pena and Dr.Moulton
Elevator Pitch
Design Approach
Several designs were considered to support the enema administration. The initial design consisted of a pair of underwear with a bell-shaped support around the anus. The bell contained a hole for the Foley catheter to insert through and slid up the Foley to seal the anus. This iteration did not provide enough surface area coverage around the anus to stop any leakage. The bell was also completely external; it did not have a cone-tip to insert slightly into the anus. The underwear was too flimsy to provide a tight support. Given these conditions, the next design consisted of a skeletal underwear design. Thin nylon straps were used to provide a tighter fit, and velcro was attached in three places to allow for adjustability. The bell was redesigned into a wedge shape. This wedge provided much more surface area around the anus, ensuring a seal. For the final design a belt and strap system with wedge support were proposed to provide the desired seal.


Design Solution
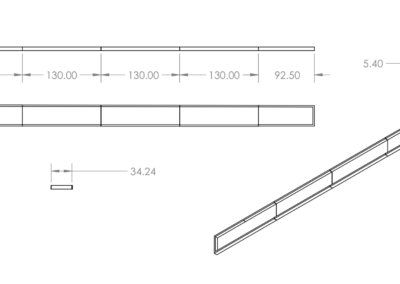
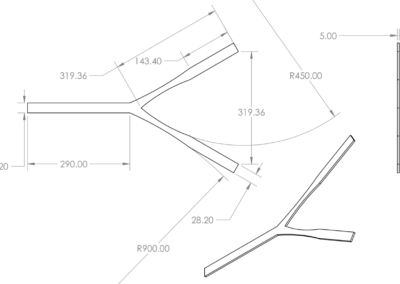
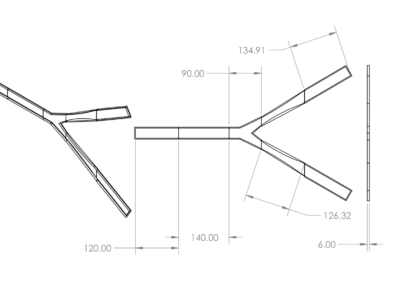
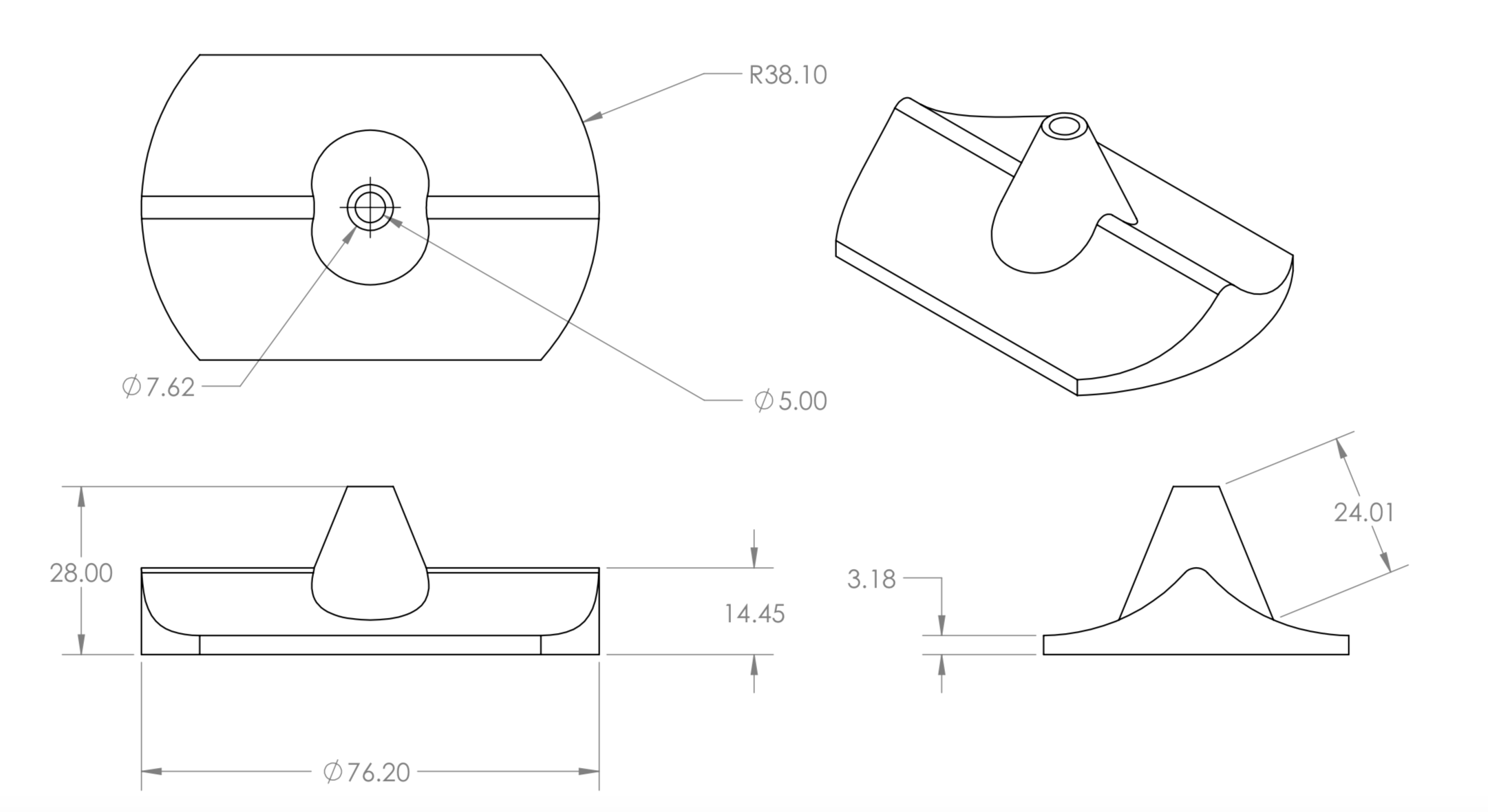
A two-strap system with external support was chosen and is shown the first figure below. It contains a silicone wedge to seal the anus and several pins to allow for adjustability. This prototype is meant for children with spina bifida, but can be altered to fit patients with various fecal incontinence issues. The dimensions for the system were determined from a mannequin torso model, and models were rendered as to be easily scaled for users of any size. The system is composed of four components including the Foley catheter. The first two components, labeled A in the figure, are a y-strap and wedge which fit under the patient and are tensioned during enema administration to provide a seal. The y-strap and wedge are composed of silicone, the Shore Hardness and tensile strength of the strap are higher than that of wedge. The Foley catheter, B in the figure, is threaded through a hole in the wedge at the beginning of the enema. The belt, C in the figure, wraps around the patient’s waist and supports the tension on the y-strap and wedge. The belt is composed of the same silicone material as the y-strap. The belt and straps are held together using connecting pins. The connecting pins are specially designed to work with the silicone material. Typical connecting pins such as buttons or Chicago screws cannot be mounted in the flexible silicone, as the torsional forces they experience when mounted are sufficient to rip the pins from the material. To reliably mount the pins in the material the pin base and silicone were made to act as interlocking toruses. It is topologically impossible for the pins to be removed from the silicone straps as long as either material does not break in such a way as to incomplete the torso. Testing of the apparatus to be carried out in future work will involve a torsional stress test on the pin design. They are currently sufficient to secure the belt and strap system snuggly around the model torso. These pins were incorporated into the third design iteration as conventional means of pin attachments failed to remain in the silicone under torsion. From the third design it was clear the straps were too thick around the genital area to mesh comfortably with the human anatomy. The fourth and final design was cast in silicone with thinner straps to correct this issue. Additionally the loop in the belt was removed to ease dressing as the pin connectors were found to be sufficient to attach the straps. The goal of the final design, shown in the second figure below, is to insure clean and complete administration of the enema. This final design aids children with spina bifida in achieving a quality of life that was previously unavailable due to the unreliability of the Foley catheter on its own. The final design of this project works synergistically with the Foley catheter to create a perfect seal around the anus. This is highly beneficial to not only keep costs low but minimize the risk of a failed enema administration and improve the quality of life for those who suffer from this condition.


Next Steps
A $20,000 grant has been received for the continuation of this project. This funding comes from the Children’s Hospital and Colorado School of Mines. The final design will be remolded in a skin-safe silicone and tested with a model for baseline data. A testing procedure has been compiled to test various performance criteria of the device. Internal pressure and leakage will be recorded on a testing vessel designed to model human anatomy. This testing vessel will be fabricated using the allocated funds. Following model testing the doctors at Children’s Hospital plan to use the enema device in medical trials. Should the device improve enema administration it will be mass manufactured and made available to patients with various rectal motility complications.


Meet the Team
Ryan Croghan

Ryan is a mechanical engineer with a focus in automation. He grew up in Weld County, CO.
Mary King

Mary is graduating with a degree in Mechanical Engineering. She has enjoyed going to Mines the last few years and will miss the mountains near Golden but is excited to move on to a career as a consultant in Houston, Texas.
Rachel McManus

Rachel McManus is a graduating mechanical engineering student originally from Colorado Springs, CO. She’s pursuing a career in aerospace or fields related to continuum mechanics (fluid mechanics, thermodynamics, and heat transfer).
James McNamara

James is a mechanical engineer with an interest in computer science. He’s hoping to find a career with overlap between the two fields.
Gabriel Muehlbauer

Gabe is getting a degree in Mechanical Engineering and would like to focus on Additive Manufacturing for a career. Gabe likes Jesus, guns, people, nature and lots more. He’s really looking forward to graduating.